This is an archived article that was published on sltrib.com in 2013, and information in the article may be outdated. It is provided only for personal research purposes and may not be reprinted.
On Mary Bronner's desk at the Huntsman Cancer Institute stands a microscope for identifying and categorizing cancer cells.
About 99 percent of the diagnostic work by her team of pathologists is done with this centuries-old tool.
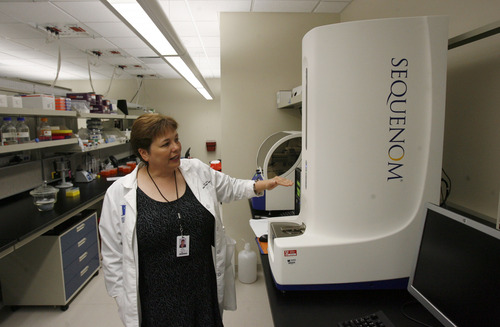
But in lab space down the hall from Bronner's office is the future: a high-powered $250,000 DNA sequencing machine that some say is revolutionizing cancer care.
Wooed by the promise of "precision" medicine — the ability to tailor treatment to a patient's genetic makeup or gauge a person's cancer risk before they get sick — cancer centers across the country are investing heavily in genetics.
Genetics is of rising importance in many medical fields. But cancer is big business and hospitals that specialize in it are well financed, able to afford the newer technologies and trained experts.
"In the U.S. alone over half-a-million people a year succumb to this disease. That's more than one person of every minute of every day," said Mary Beckerle, Huntsman's CEO. "It is a huge unmet medical need No. 1, and No. 2, we know genetics matters in cancer and we have great examples of how understanding the genetics of the disease can help guide treatment and prevention."
Next generation sequencing technologies have made it possible to quickly and inexpensively search a patient's entire genome for known or suspected trouble-maker mutations.
This doesn't happen at Huntsman, or anywhere else in Utah, and likely won't be happening anytime soon. Looking at the whole genome, 3 billion base pairs, is still a massive, needle-in-a-haystack undertaking useful in research, but not in the clinic, said Bronner, Huntsman's Chief of Anatomic Pathology.
But Huntsman is routinely screening patients for specific genetic mutations, and Intermountain Healthcare's Dixie Regional Medical Center in St. George plans to start.
This spring Huntsman started screening all patients for 50 mutations for which targeted therapies exist. Later this year, Dixie Regional will launch a similar "personalized" medicine program.
"Everyone who comes to Huntsman and has failed a standard therapy will be tested this way," said Bronner. "We're not looking at the whole genome, just a small fraction of the genome. We target them because we know we have drugs for them."
And the list of targetable genes is growing.
Just five years ago researchers made a big splash by targeting a colon cancer gene known as KRAS.
"Literally within months oncologists were demanding that every colon cancer be tested for KRAS," said Bronner. "Research has since snowballed so that we now test for four genes with every colon cancer."
Today, doctors can pinpoint and target treatments for genetic abnormalities behind all the major cancers in the United States: lung, colon, breast, ovarian, endometrial and prostate.
"This whole concept of personalized medicine is going to start taking off. It's what everyone's talking about nationally," said Lincoln Nadauld, the oncologist hired to bring cancer genomics to Dixie.
Precisely where genomics is headed, and how much promise it holds, is a matter of a debate.
Only about 5 to 10 percent of all adult cancers are hereditary, resulting directly from defect genes inherited from a parent, according to the American Cancer Society. Some scientists, though, believe more lurk undiscovered, especially in children.
Targeted therapy isn't available, or even applicable, to everyone, acknowledges Nadauld. "It isn't a magic bullet."
And insurance doesn't always cover the screening, which can range in cost from $2,000 to $16,000.
But it's a treatment option that will soon become the standard of care, predict Nadauld and Bronner. Targeted chemotherapies are easier on patients because they don't wipe out all the body's cells, and instead go after cancer cells while sparing healthy ones.
They're still very toxic drugs and they don't always work. Even though a drug should work on a particular cancer, research might show it only works 40 percent of the time, Bronner said. "There are many hypotheses as to why. Cancer is a complicated disease that involves hundreds of genes. It may wipe out part of the cancer, but it survives using an alternate pathway."
But 40 percent "is better than nothing," she said.
Sequencing also helps doctors monitor changes in tumors that cause them to become resistant to drugs, said Sunil Sharma, the hospital's chief medical oncologist and director of clinical research.
And it's used to find patients who might be good candidates for a drug trial.
"Even if they are undergoing chemo somewhere else, they can come up here and do large sequencing panel and we can advise them on experimental treatments here or elsewhere," Sharma said.
The cost of such testing is really a drop in the bucket compared to other downstream costs, said Bronner, noting it costs less than surgery or a single dose of chemotherapy.
It's too early to say whether genetic sequencing, or molecular diagnostics, will reduce those costs. Scientists at Huntsman, Intermountain and elsewhere are exploring just that.
But Bronner is a believer.
Pathology represents only 1 percent of all the cancer care costs, but all the later costs are based on the diagnosis that pathologists produce, Bronner said. "If you don't have an accurate diagnosis, all of those costs are incorrect."
Twitter: @kirstendstewart —
Genetic sequencing in research
Researchers are using genetic sequencing to speed delivery of new drugs, and to prevent disease from taking hold in patients with a family history of cancer, blurring the line between research and medicine.
Drug trials
At Huntsman Cancer Institute, researchers just wrapped up a clinical trial on a new lung cancer drug. Sequencing was used to screen 200 patients to find the 5 to 10 percent who carried the genetic change that made them a candidate for the trial.
Such studies are expanding the arsenal of available drugs, said Sunil Sharma, chief medical oncologist and director of clinical research at Huntsman. "If this drug was tested in 100 lung cancer patients it would have been deemed a failure."
Early Detection
Scientists at Huntsman took part in a national trial to monitor children with Li-Fraumeni syndrome (LFS), an inherited disorder that leads to cancer. Those with the syndrome have a 70 to 80 percent chance of developing cancer or multiple cancers in their lifetime.
Researchers devised a surveillance strategy of routine blood tests and full-body MRI scans to detect cancer in children.
After three years, 100 percent of those who submitted to screening were still alive, said Joshua Schiffman, who directs a pediatric cancer genetics clinic at Huntsman.
Those who didn't want screening had poorer outcomes. When they arrived at the clinic with cancer symptoms, only 21 percent were alive after three years, said Schiffman, who encourages and offers surveillance for all Li-Fraumeni patients.